Galvanized steel is all around us and plays a vital role in our everyday lives. It is used in construction, transport, agriculture, power transmission and everywhere that good corrosion protection and long life are essential. It, for instance, helps light our roads (lighting columns) and provide power for our homes, hospitals and offices (high voltage pylons). There are many other important industries that make use of galvanizing.
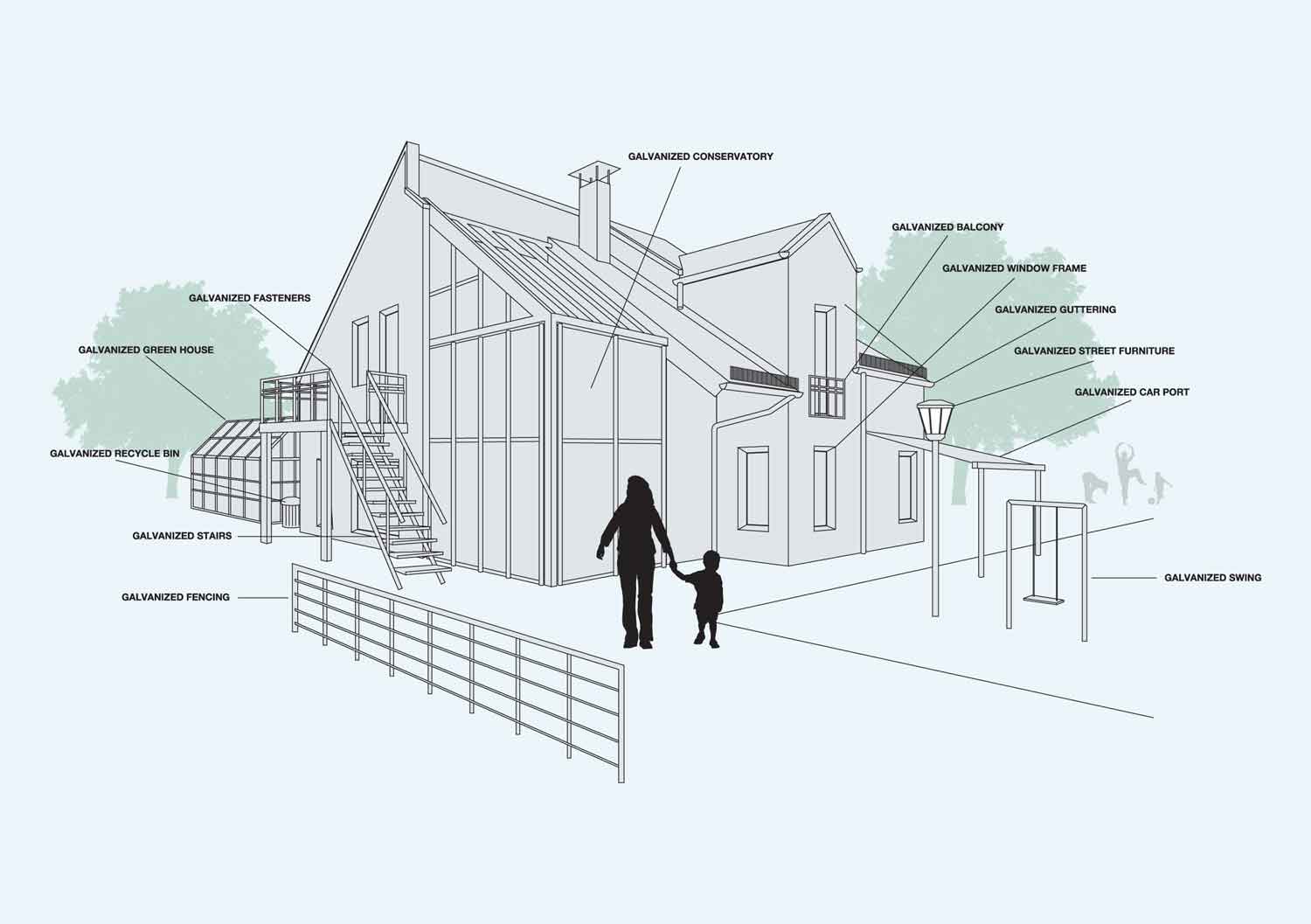 A large proportion of galvanized steel is used in construction. However, it is a very versatile process and articles ranging in size from nuts and bolts to large structural sections can be protected.
A large proportion of galvanized steel is used in construction. However, it is a very versatile process and articles ranging in size from nuts and bolts to large structural sections can be protected.
Galvanizing is a corrosion protection process for steel, in which the steel is coated with zinc to prevent it from rusting. The process involves dipping cleaned iron or steel components into molten zinc (which is usually around 450°C). A series of zinc-iron alloy layers are formed by a metallurgical reaction between the iron and zinc creating a strong bond between steel and the coating.
Low Life Cycle Energy
Galvanizing is energy efficient throughout its production and whole life cycle.
Resource Efficient
The galvanizing process uses natural resources considerately to ensure a relatively low environmental burden.
Recyclable
Whether it’s re-galvanizing, removal or reuse, galvanized steel is easily recycled. What’s more, it can also be recycled with steel scrap.
Long-life
Hot dip galvanizing protects against corrosion and prolongs the life of steel, greatly lowering the environmental and economic cost.
Cost Efficient
The initial investment of using galvanized steel for long-term protection in construction can provide cost benefits for decades.
Galvanized steel in Southwold
Two holiday homes on the Suffolk Coast that have replaced a derelict building can be moved as the coast retreats and thus react to climate change. The buildings are entirely recyclable with aluminium and wood windows, galvanized steel, timber frame and plaster free. Watch the video to find out more.
To find out more about galvanizing and sustainability from our technical experts, book our RIBA approved ‘Galvanizing and Sustainable Construction CPD‘.
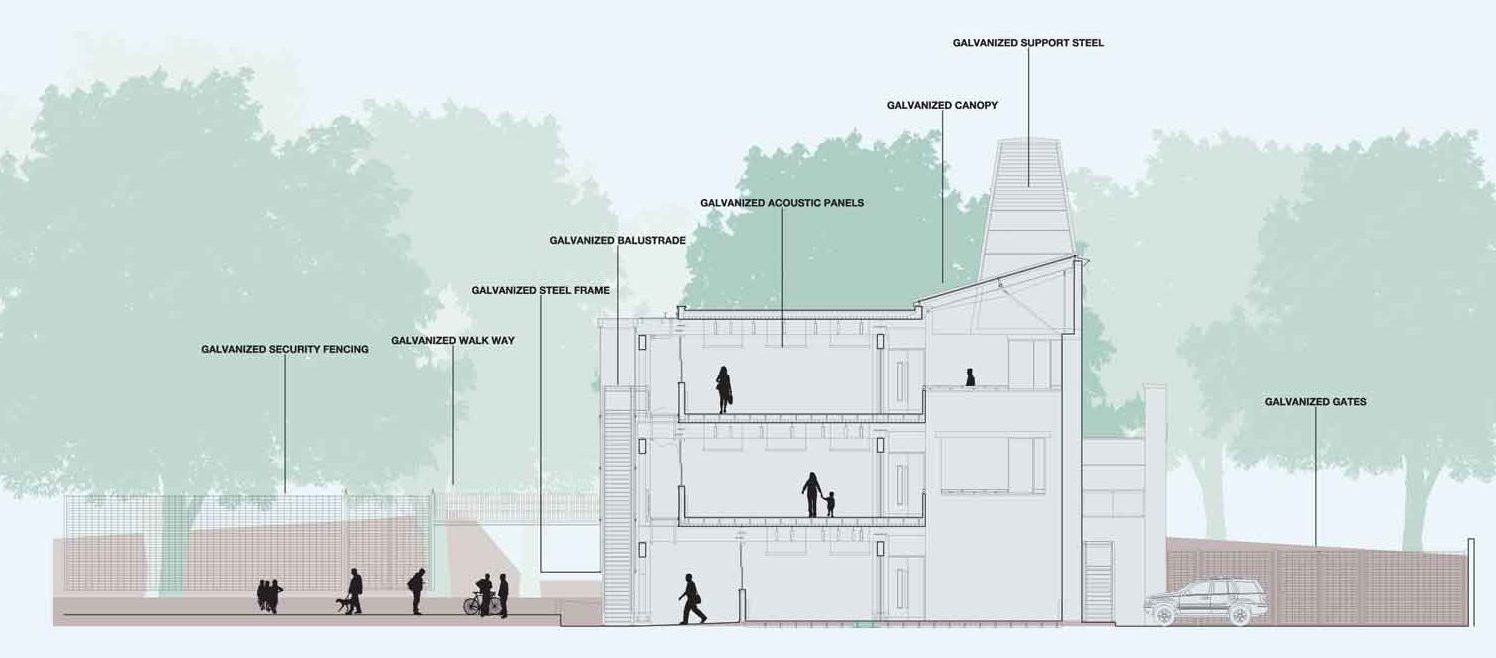
Galvanized steel – embodied carbon
Attention to durability of steel structures and components has important environmental, economic and social consequences. Some of these are less obvious than others.
 The overall economic cost of corrosion has been studied in several countries. It is commonly estimated at up to 4% of gross domestic product.
The overall economic cost of corrosion has been studied in several countries. It is commonly estimated at up to 4% of gross domestic product.
The long-term durability provided by galvanizing is achieved at relatively low environmental burden in terms of energy and other globally relevant impacts, especially when compared to the energy value of the steel it is protecting.
A review of available life cycle studies by Life Cycle Engineering (Torino, Italy) has indicated the typical metrics shown in the table below. The range represents variations in type of steel component, geographical factors and study methodology.

Using this knowledge of the environmental burdens of corrosion protection with galvanizing, it has been possible to compare the consequences of the different choices of corrosion protection systems. Several studies have demonstrated the high economic and environmental costs associated with the repeated maintenance painting of steel structures. These burdens can be significantly reduced by an initial investment in long-term protection.
Environmental aspects
Galvanizing process
Galvanizing is always carried out in an industrial works which contains all stages of the process. Steel comes in at one end and the finished galvanized product goes out at the other. There is a good spread of galvanizing plants across the UK and Ireland and steel does not have to travel great distances to a nearby galvanizing plant keeping transport costs and environmental impacts as low as possible. The main process consumable, zinc, is used very efficiently in the process. The dip operation ensures that any zinc that is not deposited on the steel is returned to the galvanizing bath. Zinc that oxidizes on the surface is removed as an ash and is readily recycled (sometimes on site). Dross formed at the bottom of the bath is removed periodically and has a high market value for recycling.
THE GALVANIZING PROCESS:
Inputs, emissions, wastes and recycling flows
Click here to view larger image.
Process energy use
Energy is required to heat the hot dip galvanizing bath and this is usually supplied by natural gas. Although the galvanizing industry is not considered to be amongst the most energy-intensive sectors of industry, it has made great efforts to manage its energy use efficiently. The galvanizing industry has set targets for energy efficiency and encouraged improved energy management and new technology to achieve these targets.
Examples of these advances are:
- improved burner technology for greater energy efficiency
- more efficient bath lids (used during maintenance and/or down time)
- greater use of waste heat for heating of pre-treatment tanks
Emission control
Emissions within the plant are carefully controlled to avoid disturbance or problems for the surrounding neighbourhood. Galvanizing plants are regulated under the EU Directive on Integrated Pollution, Prevention and Control. The industry has cooperated in the publication of a Best Practice Reference Note (BREF) for hot dip galvanizing.
The principal requirement of the BREF is to capture the non-hazardous particulates during dipping. These particulates are then filtered using either scrubbers or bag filters.
Regeneration and recycling
Pretreatment steps in the process are mainly aimed at cleaning the steel articles. Process consumables, such as hydrochloric acid and flux solutions all have important recycling and/or regeneration routes.
For example:
- spent hydrochloric acid solutions are used to produce iron chloride for use in treating municipal waste water. Many plants remove iron and zinc and recycle regenerated acid to the re-treatment tanks
- improved monitoring and maintenance of flux tanks means that these are rarely discarded to waste and only small volumes of sludge require periodic disposal. Closed-loop flux recycling is used in many plants
- ambient temperature acidic and biological degreasers have been developed
Water use
Galvanizing plants use relatively low volumes of water compared to other coating technologies. In fact, it is very rare for a galvanizing plant to discharge waste water. Any waste water that is generated can be treated and returned to the process, with only low volumes of stable solids sent for external disposal.
In some cases, it has been possible for galvanizing plants to eliminate the use of mains water by harvesting rainwater falling on the site. Rainwater can be collected through gutters and stored for later use.
Galvanizing recycling
There are two important sources of zinc used in the galvanizing process:
- refined zinc is produced from a mix of both mined ores and recycled feedstocks. It is estimated that, on average, refined zinc contains about 10-15% of recycled feedstock
- galvanizers are also important purchasers of remelt zinc – that is scrap zinc from, for example, old zinc roofs that have been cleaned and remelted into ingot form
So, the refined zinc purchased by galvanizing plants contains a high proportion of recycled zinc and fully recycled zinc is often purchased to supplement use of refined zinc.
During the galvanizing process, any zinc that does not form a coating on the steel remains in the bath for further reuse. There is no loss of materials that may occur during spray application of other coating types.
Zinc ash (from surface oxidation of the galvanizing bath) and dross (a mix of zinc and iron that accumulates at the bottom of the galvanizing bath) are fully recovered. Any zinc metal within the crude ash is directly recycled for further use, often in the same galvanizing process.
The fine ash and dross are then sold to make zinc dust and compounds for a variety of applications such as rubber additives, cosmetics and electronic components.
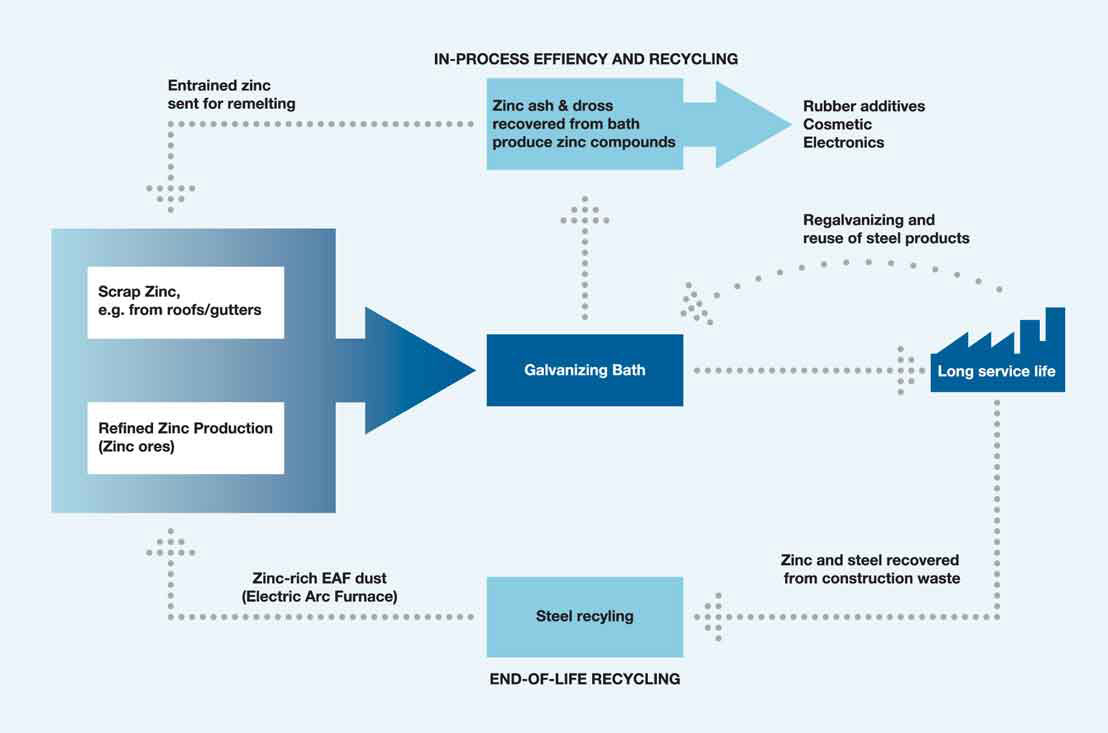
Galvanized steel can be recycled easily with other steel scrap in the electric arc furnace (EAF) steel production process. Zinc volatilises early in the process and is collected in the EAF dust that is then recycled in specialist facilities and often returns to refined zinc production.
In 2006, the European steel industry (EU27) produced 1,290,750 tonnes of EAF dust, which contained 296,872 tonnes of zinc (i.e., 23%). 93% of this zinc (276,920 tonnes) was recycled. (source: Gesellschaft für Bergbau, Metallurgie, Rohstoff – und Umwelttechnik, Germany).
Steel products often have a very long useful life, e.g. many very old steel bridges are still in use. For that reason there is a shortage of scrap and the constant growth in infrastructure will have to be based on primary production of iron ore. The same applies to many other metals that are used in applications with a long useful life.
Durability and service life
Hot dip galvanizing to EN ISO 1461 ensures a coating of zinc is applied to protect steel.
This is important to provide extended durability, especially in outdoor environments. Thinner zinc coatings will not last so long because the life of a zinc coating is directly proportional to its thickness.
Zinc’s resistance to corrosion depends primarily on a protective film (patina) formed on its surface. For atmospheric corrosion, atmospheric contaminants affect the nature and durability of this film. The most important contaminant affecting zinc is sulphur dioxide (SO2) and it is the presence of SO2 which largely controls zinc’s atmospheric corrosion rate.
 It is widely documented that atmospheric SO2 levels have fallen significantly in most countries over the past decades.
It is widely documented that atmospheric SO2 levels have fallen significantly in most countries over the past decades.
The link between zinc durability and atmospheric SO2 levels has been illustrated very clearly for Sweden. Similar data have been recorded for other countries.
This downward trend and consequent improvements in zinc performance significantly enhance the contribution of galvanized steel to sustainable development by providing, at no additional cost, more durable structures for shelter, infrastructure, transportation and many other applications.
With zinc corrosion rates normally less than 1µm per year, a typical 85µm coating can provide many decades of maintenance-free life (in rural and urban environments).
For more aggressive environments, it is possible to achieve thicker coatings on structural steelwork with corresponding increases to coating life.


